BIN1 Antibody
Mouse Monoclonal 99F IgG
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
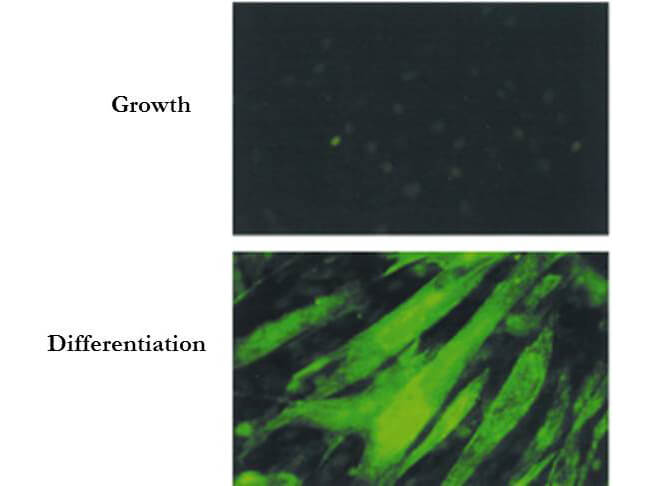
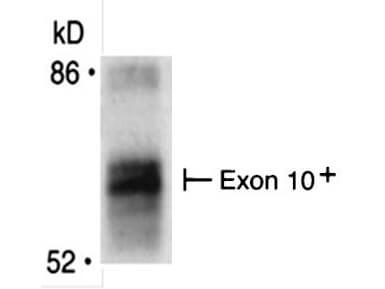
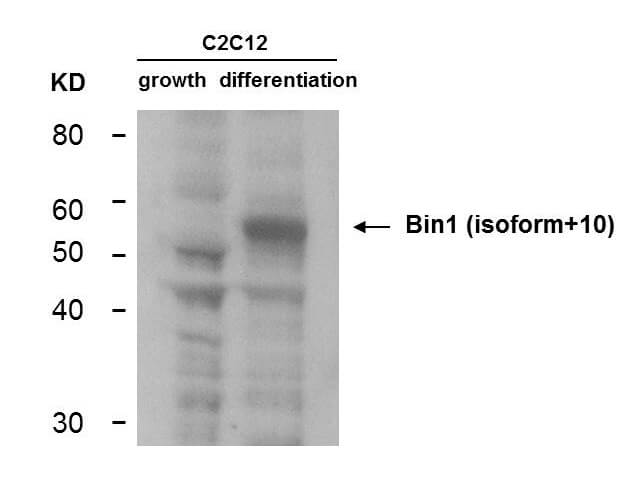
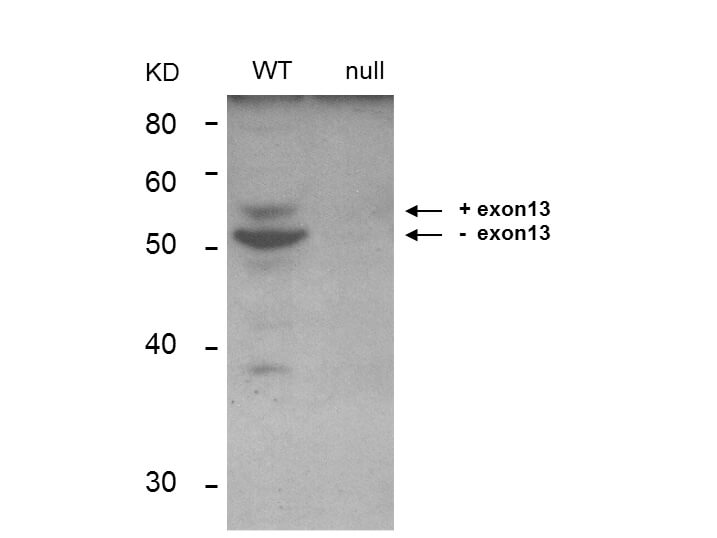
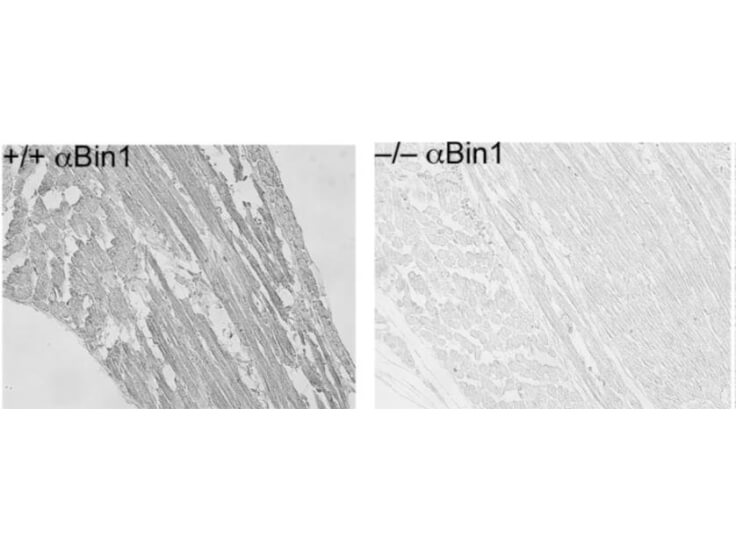
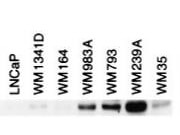
Bin1 is a conserved member of the BAR family of genes that have been implicated in diverse cellular processes including endocytosis, actin organization, programmed cell death, stress responses, and transcriptional control. The first mammalian BAR protein to be discovered, Amphiphysin I (AmphI), was identified in an immunoscreen for proteins associated with the plasma membranes of synaptic neurons, functions in the control of clathrin-dependent synaptic vesicle endocytosis. The mammalian Bin1 gene was first identified in a two hybrid screen for polypeptides that bind to the N-terminal Myc box 1 (MB1) portion of the c-Myc oncoprotein. Bin1 is similar to AmphI in overall structure, with an N-terminal BAR domain and a C-terminal SH3 domain. However, the Bin1 gene is more complex than the AmphI gene, encoding at least seven different splice variants that differ widely in subcellular localization, tissue distribution, and ascribed functions. Alternate splicing of the Bin1 gene results in ten transcript variants encoding different isoform. Bin1 is expressed ubiquitously in mammalian cells. Certain splice variants of Bin1 are expressed in the neurons, muscle cells or tumor cells and play a role in cancer suppression. Studies in muscle cells suggest that Bin1 expression, structure, and localization are tightly regulated during muscle differentiation and suggested that Bin1 plays a functional role in the differentiation process. Defects in BIN1 are the cause of centronuclear myopathy autosomal recessive; also known as autosomal recessive myotubular myopathy.
Product Details
Target Details
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.