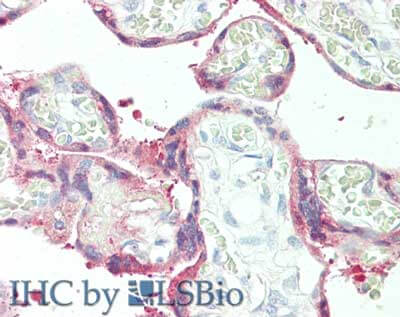
Plasminogen Activator Inhibitor 2 Antibody
Rabbit Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
Extracellular plasminogen activator inhibitor type-2 (also known as PAI-2, Placental plasminogen activator inhibitor; Monocyte Arg-serpin; Urokinase inhibitor; SERPINB2 serpin peptidase inhibitor, clade B (ovalbumin), member 2) is a coagulation factor and a potent inhibitor of urokinase-type plasminogen activator (u-PA) and also acts as a multifunctional protein. It is present in most cells, especially monocytes and macrophages. PAI-2 exists in two forms, a 60-kDa, secreted, extracellular, glycosylated form and a 43-kDa intracellular form. It is a multifunctional protein that plays a role in cell differentiation, in prevention of programmed cell death, in the regulation of cell proliferation, in the inhibition of microbial proteinases and in the protection against stromal degradation. High levels of the PAI-2 protein are associated with a good prognosis in breast cancer, small cell lung, ovarian cancer, and inhibition of metastasis. PAI-2 also plays a role in inflammation on the surface of the eye. PAI-2 may cooperate with pRb2/p130 in modulating PAI-2 gene expression by chromatin remodeling.
Product Details
Target Details
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.