Rad9 phospho S1260 Antibody
Rabbit Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
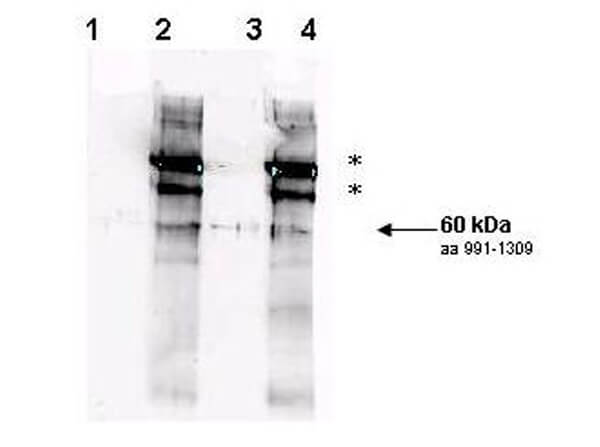
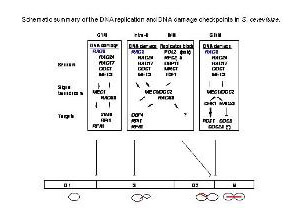
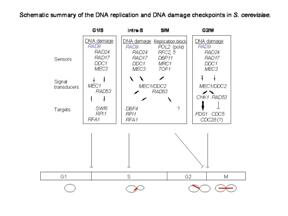
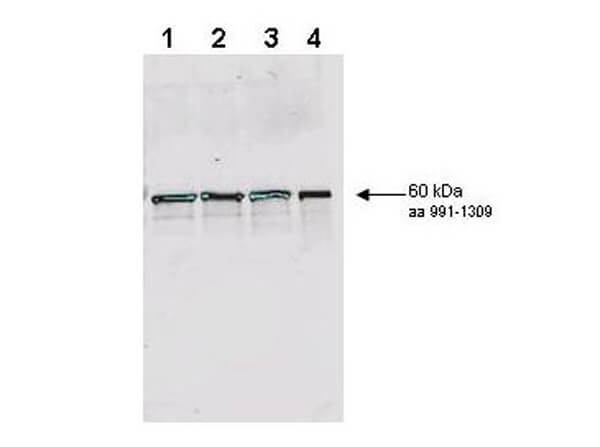
Rad9 is required for the MEC1/TEL1-dependent activation of Saccharomyces cerevisiae DNA damage checkpoint pathways mediated by Rad53 and Chk1. DNA damage induces Rad9 phosphorylation, and Rad53 specifically associates with phosphorylated Rad9. Cells have evolved multiple strategies for tolerating genomic damage. The most important of these are numerous repair systems that remove or bypass potentially mutagenic DNA lesions. Another cellular strategy is to delay cell-cycle transitions at multiple points. The genetic control of these delays, termed `checkpoints', was first established in budding yeast where it was shown that the RAD9 gene functions in G2/M arrest after irradiation with X-rays. Subsequently, it has become clear that Rad9 also functions at the G1/S, intra-S and mid-anaphase checkpoints. Defects in checkpoint regulation can lead to genome instability and, in higher eukaryotes, neoplastic transformation. Rad9 also controls the transcriptional induction of a DNA damage regulon (DDR). Rad9 may also have a pro-apoptotic function. This is suggested in that Rad9 from Schizosaccharomyces pombe (SpRad9) contains a group of amino acids with similarity to the Bcl-2 homology 3 death domain, which is required for SpRad9 interaction with human Bcl-2 and apoptosis induction in human cells. Overexpression of Bcl-2 in S. pombe inhibits cell growth independently of rad9, but enhances resistance of rad9-null cells to methyl methanesulfonate, ultraviolet and ionizing radiation. Rad9 conveys the checkpoint signal by activating Rad53p and Chk1p; is hyperphos-phorylated by Mec1p and Tel1p; and is a potential Cdc28p substrate. Mature yeast Rad9 is reported to have an apparent molecular weight of ~148kDa. The human homolog is reported at 48.5 kDa.
Product Details
Target Details
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.