CHK2 phospho T68 Antibody
Rabbit Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
CHK2 (also known as CHEK2, Protein kinase CHK2 isoform a, and checkpoint-like protein) is a serine/ threonine-protein kinase involved in the control of cell cycle checkpoints and may also participate in transduction of the DNA damage and replicational stress signals. CHK2 is the mammalian ortholog of the budding yeast Rad53 and fission yeast Cds1 checkpoint kinases. The amino-terminal domain of CHK2 contains a series of seven serine and threonine residues (Ser19, Thr26, Ser28, Ser33, Ser35, Ser50 and Thr68) followed by glutamine (SQ or TQ motif). These are known to be preferred sites for phosphorylation by ATM/ATR kinases. Indeed, after DNA damage by ionizing radiation (IR), UV irradiation or hydroxyurea treatment, Thr68 and other sites in this region become phosphorylated by ATM/ATR. The SQ/TQ cluster domain, therefore, seems to have a regulatory function. Phosphorylation at Thr68 is a prerequisite for the subsequent activation step, which is attributable to autophosphorylation of Chk2 on residues Thr383 and Thr387 in the activation loop of the kinase domain. CHK2 inhibits CDC25C phosphatase by phosphorylating it on Ser-216, preventing the entry into mitosis. This kinase may have a role in meiosis as well. Kinase activity is up regulated by autophosphorylation and the protein is rapidly phosphorylated in response to DNA damage and to replication block.
Product Details
Target Details
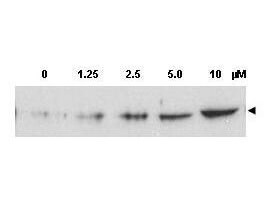
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.