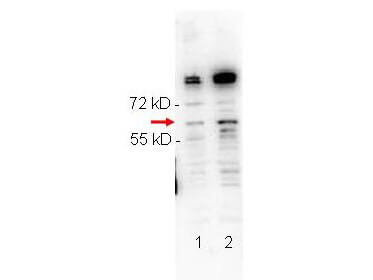
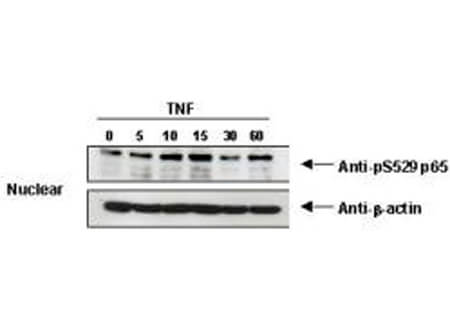
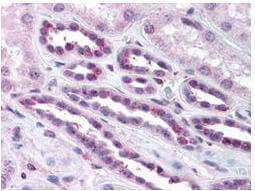
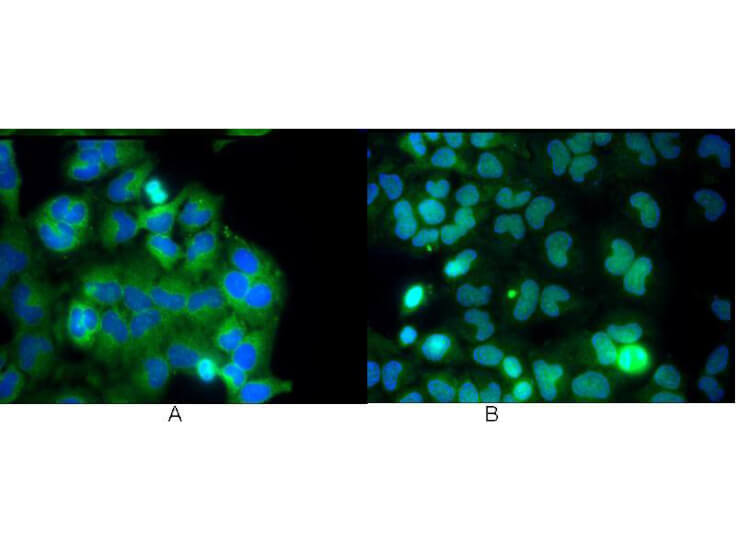
NFkB p65 phospho S276 Antibody
Rabbit Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
NFkB was originally identified as a factor that binds to the immunoglobulin kappa light chain enhancer in B cells. It was subsequently found in non-B cells in an inactive cytoplasmic form consisting of NFkB bound to IkB. NFkB was originally identified as a heterodimeric DNA binding protein complex consisting of p65 (RelA) and p50 (NFKB1) subunits. Other identified subunits include p52 (NFKB2), cRel, and RelB. The p65, cRel, and RelB subunits are responsible for transactivation. The p50 and p52 subunits possess DNA binding activity but limited ability to transactivate. p52 has been reported to form transcriptionally active heterodimers with the NFkB subunit p65, similar to p50/p65 heterodimers. Low levels of p52 and p50 homodimers can also exist in cells. The heterodimers of p52/p65 and p50/p65 are regulated by physical inactivation in the cytoplasm by IkB-alpha. IkB-alpha binds to the p65 subunit preventing nuclear localization and DNA binding. Activators mediate a rapid phosphorylation of IkB by IkB kinase (IKK) which results in subsequent ubiquitination and proteolytic degradation. NFkB is then transported to the nucleus, where it activates transcription of target genes through binding to NFkB target sequences within the promoter. The HTLV-I protein Tax can induce constitutive NFkB activation through phosphorylation of both IkB-alpha and IkB-beta. The transforming protein Tax inhibits p53 transcriptional activity through the NFkB signaling pathway, specifically via the p65 (RelA) subunit.
Product Details
Target Details
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.