p39 Antibody
Rabbit Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
The p39 protein, or Basic membrane protein A, is one of the immunogenic cell membrane components of Borrelia burgdorferi, the spirochete carried by Ixodes ticks. The spirochete migrates from the tick midgut during feeding to its salivary glands and are thus transmitted to the mammal host. This transition may be facilitated by changes in expression of some B. burgdorferi genes. It is believed that expression of the various proteins associated with the spirochete may be regulated by the changes in tick life cycle, changes in conditions during tick feeding (such as temperature, pH, and nutrients) and/or in coordination with the course of infection of the mammal host. BmpA is expressed during the invasion of the spirochete and in the evolution of the arthritis of Lyme disease in mammals. It belongs to the BMP lipoprotein family. The major products of the B. burgdorferi basic membrane protein (bmp) A/B operon that are induced in murine and human joints possess inflammatory properties. Non-lipidated and lipidated versions of BmpA have been shown to induce the pro-inflammatory cytokine TNF-α and IL-1ß in human synovial cells. The induction of cytokine responses in synovial cells via activation of the NF-kappaB and p38 MAP kinase pathways could potentially contribute to the genesis of Lyme arthritis. The BmpA outer-surface protein is an important antigen for serodiagnosis of human infection. B. burgdorferi adheres to host extracellular matrix components, including laminin, but may not bind mammalian type I or type IV collagens or fibronectin.
Product Details
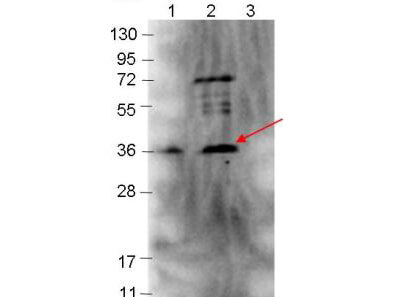
Target Details
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.