PI3-kinase p110 delta Antibody
Rabbit Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
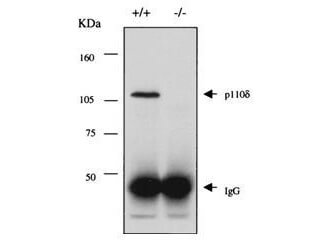
Phosphoinositide 3-kinases (PI3Ks) generate 3-phosphoinositide lipids in cell membranes. A variety of intracellular target proteins interact with these lipids via specific lipid-binding modules and, as a consequence, undergo changes in their localization and/or activity. In this way, PI3Ks participate in the regulation of mitogenesis, differentiation, survival, intracellular vesicular transport, cytoskeletal reorganization, and motility. Tyrosine kinases and Ras use PI3Ks as essential intracellular signal relay molecules. PI3Ks are heterodimeric enzymes consisting of a regulatory subunit in complex with a p110 catalytic subunit. Mammmals have genes encoding three distinct catalytic subunits (p110α, p110ß, and p110δ and three regulatory subunits (p85α, p85ß, and p55δ). All of the p110 isoforms are capable of interacting with each type of regulatory subunit. They are also similarly recruited to phosphotyrosine complexes and have, at least in vitro, the same lipid substrate specificity. However, it is becoming increasingly clear that PI3K isoforms differ in their interaction with Ras and regulation of lipid kinase activity, and in their protein kinase activities. Several groups have provided evidence that p110 isoforms have nonredundant functions in the regulation of cell proliferation, survival, actin cytoskeleton reorganization, and migration downstream of given receptors. This antibody is specific for the carboxy terminal end of p110d that is expressed predominantly in leukocytes.
Product Details
Target Details
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.