ERK2 Antibody
Rabbit Polyclonal
$50.00 to US & $70.00 to Canada for most products. Final costs are calculated at checkout.
Background
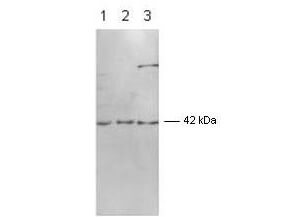
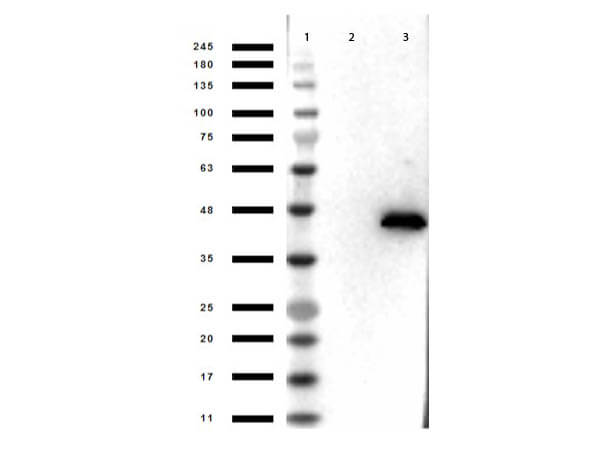
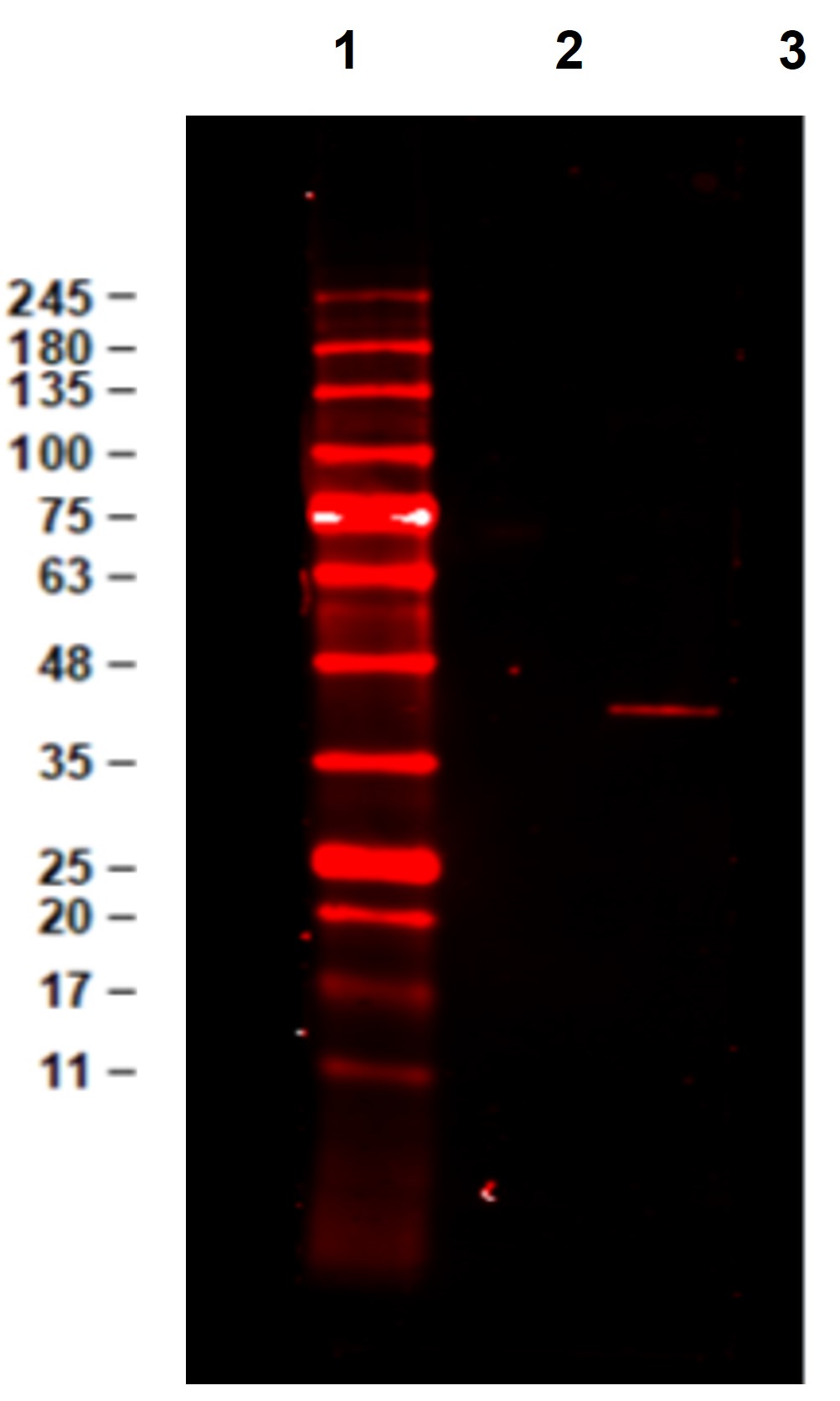
Cell proliferation is regulated in several contexts, for example during development, tissue differentiation, wound healing and immune responses. In mammalian cells, proliferative signals lead to the activation of a protein kinase cascade, resulting in the phosphorylation of two closely related Mitogen-Activated Protein Kinases (MAPK’s) ERK1 and ERK2 of 44 kDa and 42 kDa, respectively. When activated, ERK’s form dimers that translocate to the nucleus where they phosphorylate several classes of transcription factors which are involved in the up-regulation of immediate early genes. As such, ERK1 and ERK2 represent a paradigm for a growing family of proline-directed protein kinases that mediate entry, progression and exit from the cell cycle in diverse eukaryotic cells. These enzymes function within highly conserved cascade of sequentially activating protein kinases that transduce signals from diverse extracellular stimuli. Alternative splice transcript variants encoding different protein isoforms have been described. ERK1 and ERK2 are phosphorylated within the activation loop on both a Threonine and a Tyrosine residue (within a Thr-Glu-Tyr motif) by MEKs (MAPK/ERK kinases), thereby greatly elevating the activity of ERK1&2. In vertebrates the mitogen-induced sequential activation of the kinases Raf1->Mek1->Erk2->Rsk occurs via the G-protein Ras.
Product Details
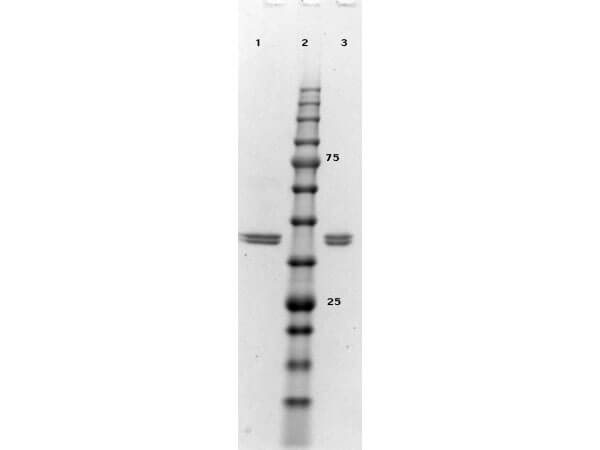
Target Details
Application Details
Formulation
Shipping & Handling
This product is for research use only and is not intended for therapeutic or diagnostic applications. Please contact a technical service representative for more information. All products of animal origin manufactured by Rockland Immunochemicals are derived from starting materials of North American origin. Collection was performed in United States Department of Agriculture (USDA) inspected facilities and all materials have been inspected and certified to be free of disease and suitable for exportation. All properties listed are typical characteristics and are not specifications. All suggestions and data are offered in good faith but without guarantee as conditions and methods of use of our products are beyond our control. All claims must be made within 30 days following the date of delivery. The prospective user must determine the suitability of our materials before adopting them on a commercial scale. Suggested uses of our products are not recommendations to use our products in violation of any patent or as a license under any patent of Rockland Immunochemicals, Inc. If you require a commercial license to use this material and do not have one, then return this material, unopened to: Rockland Inc., P.O. BOX 5199, Limerick, Pennsylvania, USA.